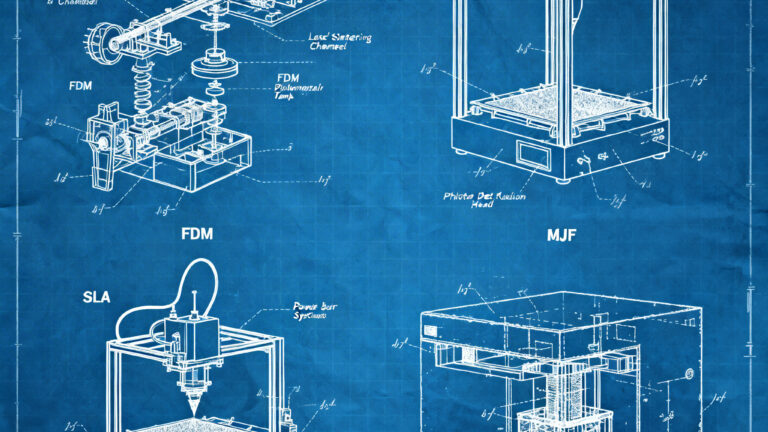
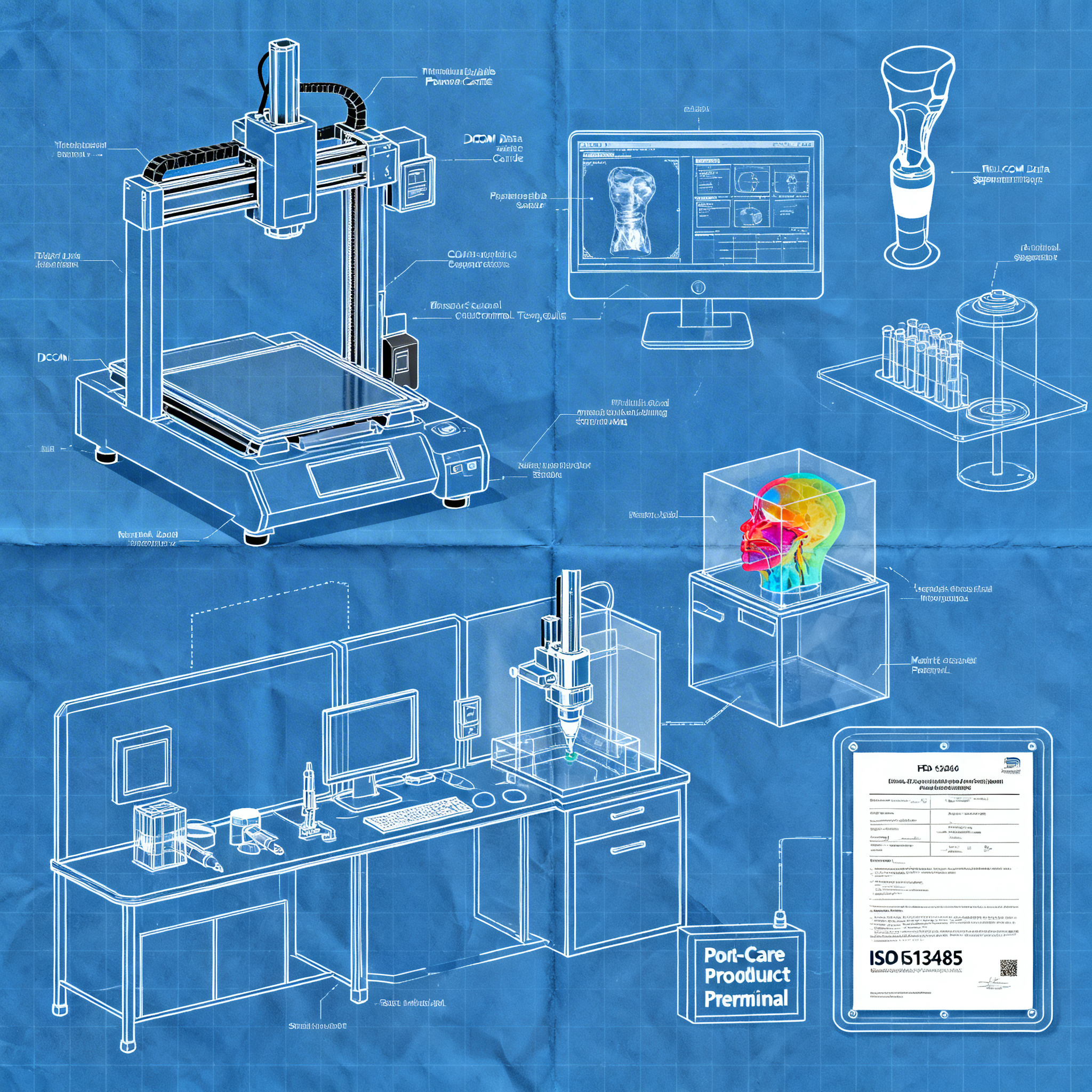
Metrology in 3D Scanning: How Integrated Processing Works and Its Industrial Applications
Integrated metrology in 3D scanners enables real-time quality control, reducing time and costs. Thanks to advanced sensors, geometric algorithms, and connectivity, these systems process dimensional data directly during scanning, integrating with metrology platforms and production processes. Despite some technological limitations, they represent a breakthrough for high-