Integration of 3D Printing in Hospitals: An Operational Guide for Safe and Efficient Clinical Implementation
It is no longer just about sporadic applications: the integration of 3D printing in hospitals requires a clear strategy on technologies, workflows, and staff training. More and more healthcare institutions are creating internal labs and developing specialized skills, transforming additive manufacturing from an experimental tool into an essential operational resource for personalized medicine.
The trend is clear: hospitals like AP-HP in France, and the Mayo Clinic and Cleveland Clinic in the United States have already integrated 3D printing into their daily clinical processes, producing custom devices, anatomical models, and tailored surgical tools internally. This point-of-care approach reduces waiting times, improves clinical precision, and, in the long term, generates significant savings compared to outsourcing.
Technology Selection and Biocompatible Materials
The choice of printing technology and the right materials is fundamental to ensuring safety and clinical compliance, with particular attention to biocompatibility and regulatory standards.
The first strategic decision concerns which technology to adopt based on the expected clinical applications. For custom titanium implants, such as the cranial prostheses produced by AP-HP, metal additive manufacturing printers capable of working with certified biocompatible materials are necessary. For pre-operative anatomical models, technologies like material jetting and PolyJet offer unique advantages: they can replicate the mechanical properties of tissues, include multiple colors, and create transparent or semi-transparent structures that allow for the visualization of tumors and neural tracts within the brain parenchyma.
Biocompatibility is a non-negotiable requirement for any device intended for patient contact. Materials must be certified according to international standards and comply with EU Regulation 2017/745 on medical devices in Europe, while in the United States, the FDA has published specific technical guidelines for devices made with additive manufacturing. Even for non-implantable devices such as surgical guides, positioning tools, and operating room accessories, material traceability and process control remain essential.
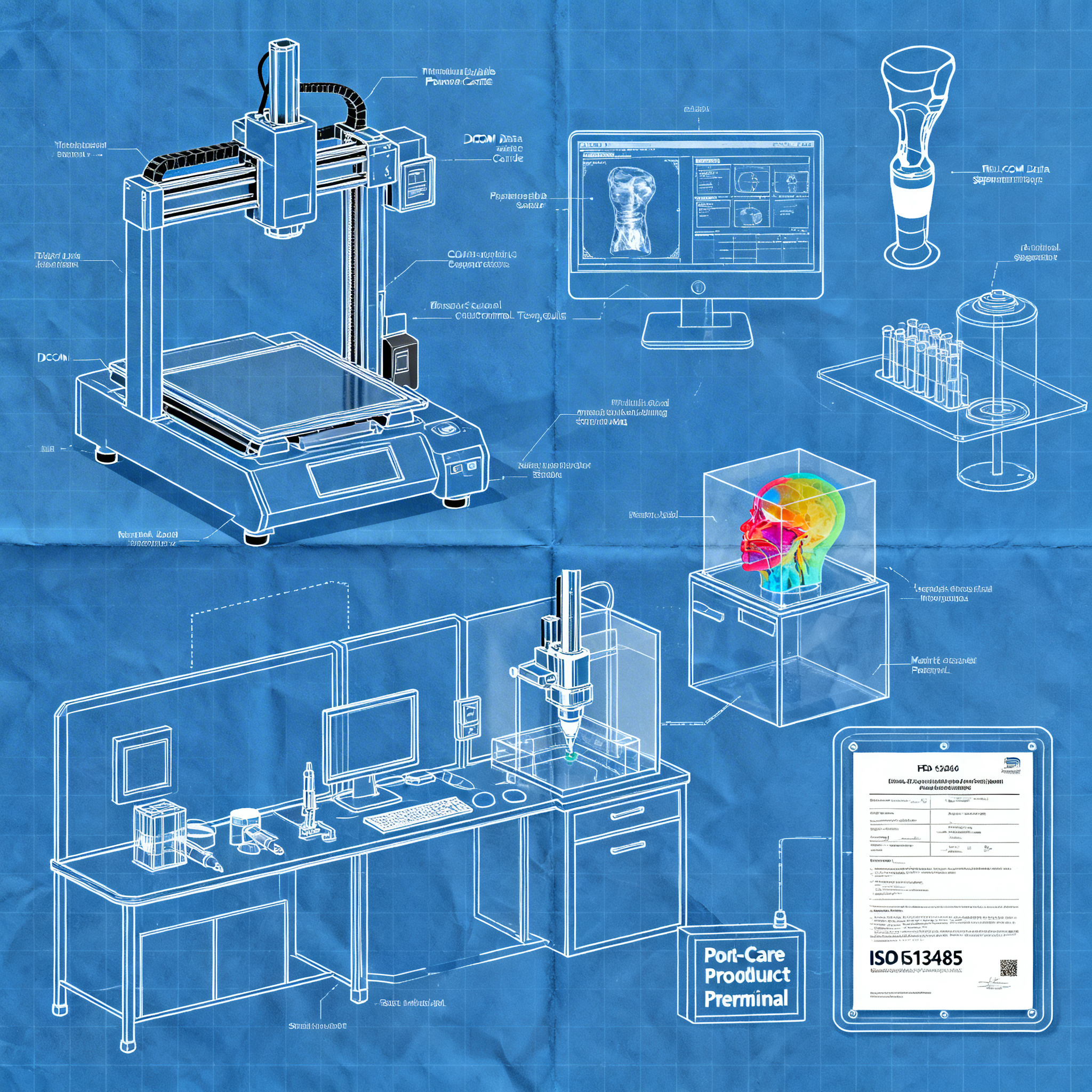
DICOM-to-Print Workflow: From Image to Production
A fluid and standardized process from imaging to printing is essential to maintain quality and acceptable clinical times, ensuring anatomical accuracy and repeatability.
The workflow begins with the acquisition of medical images through techniques such as magnetic resonance imaging (MRI), computed tomography (CT), or diffusion tensor imaging (DTI). These data, typically in DICOM format, are sent to the 3D printing laboratory through dedicated case management portals. The segmentation phase is critical: using specialized software, anatomical structures are isolated from imaging data, either by manually tracing structures along sections or by using thresholding tools when anatomy clearly contrasts with surrounding tissues.
Medical collaboration is fundamental in this phase. For example, in the case of complex brain tumors, neurosurgeons must identify critical structures such as the optic radiations, corticospinal tract, and language pathways that may not be immediately visible in imaging data. This collaboration between clinicians and 3D printing technicians allows for the creation of 1:1 models with color coding that visualize the spatial relationships between the tumor and surrounding functional tracts.
The “one-stop” model described in some hospital implementations covers the entire cycle: from conceptualization and visualization to prototyping and final production, with applications in cardiology, vascular surgery, and oncology. The “few steps away” logic allows departments to bring practical problems and have them evolve rapidly through prototype-test-correction cycles.
Staff Training and Laboratory Structuring
Integration requires trained professional figures and dedicated spaces to ensure efficiency and regulatory compliance, with skills ranging from imaging to post-production.
Who operates the machines? The answer varies, but a hybrid model emerges: specialized additive manufacturing technicians work closely with clinical staff trained on specific applications. Some hospitals integrate biomedical engineers, radiology technicians, and even surgeons with 3D modeling skills. Training must cover not only machine operation but also material management, post-processing, acceptance criteria, and full traceability of the production process.
Laboratory structuring requires dedicated spaces with adequate environmental control, quality management systems compliant with standards such as ISO 13485, and standardized operating procedures for each production phase. According to documented experiences, the most effective organizational choice is to position competencies and tools where clinical needs arise, reducing friction and increasing the ability to realize custom solutions.
An often-overlooked aspect is the creation of collaborative networks: some projects demonstrate how 3D files and production guidelines can be shared among different healthcare centers, allowing peripheral hospitals and training centers to locally print their own simulators and devices. Production costs per single module can be reduced to a few tens or hundreds of euros, compared to the thousands required for commercial equivalents. The Mayo Clinic has documented that producing anatomical models internally costs from 10% to 50% less than outsourcing.
Conclusion
The integration of 3D printing in hospitals is now a consolidated operational reality, but it requires a systematic and well-planned approach that considers technology, processes, and people as interconnected elements of a comprehensive strategy.
Beyond the technology itself, an entire care model is evolving: more personalized, more responsive, and closer to the patient. The ability to produce custom devices, prepare complex interventions with accurate anatomical models, and reduce operational costs represents a significant competitive advantage for healthcare institutions. In the long term, hospitals will no longer be just places of care, but also centers of medical production and innovation, where bioprinting and 3D drug printing will become daily practices.
Start evaluating the technological and organizational requirements to introduce 3D printing solutions in your clinical environment. Consider which clinical applications could benefit most from personalization, identify available internal skills or those to be developed, and plan a gradual implementation path that ensures regulatory compliance and economic sustainability. Tomorrow's medicine is being built today, one layer at a time.
article written with the help of artificial intelligence systems
Q&A
- What are the main benefits of integrating 3D printing in hospitals?
- 3D printing in hospitals enables point-of-care production of customized devices, anatomical models, and surgical tools. It reduces wait times, improves clinical precision, and generates cost savings compared to outsourcing.
- What 3D printing technologies are used in the clinical setting and what are they used for?
- For biocompatible metal implants, metal additive manufacturing printers are used, while for anatomical models, technologies like material jetting and PolyJet are preferred. The latter allow for the replication of tissue mechanical properties and the creation of transparent or colored structures for better anatomical visualization.
- How is safety and regulatory compliance ensured in medical 3D printing?
- Materials must be biocompatible and certified according to international standards such as EU Regulation 2017/745 or FDA guidelines. Material traceability, production process control, and the adoption of quality management systems like ISO 13485 are also essential.
- What is the role of the DICOM-to-print workflow in the production of 3D models?
- The workflow starts with the acquisition of DICOM images via MRI, CT, or DTI, continuing with segmentation assisted by specialized software. Collaboration between technicians and clinicians is crucial to correctly isolate anatomical structures and create precise, clinically useful models.
- What skills and professional profiles are needed to implement a hospital 3D printing laboratory?
- Specialized technicians in additive manufacturing, biomedical engineers, radiology technicians, and trained clinical staff are required. Training must cover machine operation, material management, post-processing, and process traceability.
- How does 3D printing affect hospital costs and efficiency?
- In-house production can reduce costs by 10-50% compared to outsourcing. Furthermore, thanks to the 'few steps away' logic, departments can resolve issues quickly through prototype-test-correction cycles, improving efficiency and clinical response times.