Medical Bioprinting with Chitosan: Design and Implementation of Personalized Therapies for Chronic Ulcers
Chitosan bioprinting is opening new therapeutic pathways for chronic ulcers, enabling the production of customized and multifunctional patches directly at the point of care.
Chronic ulcers represent one of the most complex challenges in wound care, especially in patients with diabetes or reduced mobility, where poor blood perfusion slows tissue regeneration and favors bacterial infections. A group of researchers from the University of Mississippi has developed a new concept of a 3D-printed chitosan-based patch that does not merely cover the wound but actively supports its healing, combining barrier function, controlled release of antibacterial agents, and scaffolds for tissue regeneration. The results were published in the European Journal of Pharmaceutics and Biopharmaceutics, fitting into a broader framework of international research on chitosan and 3D printing for wound care.
Properties of Chitosan in Tissue Engineering
Chitosan offers a unique combination of biocompatibility, biodegradability, and the ability to promote cell adhesion, with an intrinsic antibacterial effect that makes it ideal for the treatment of complex wounds.
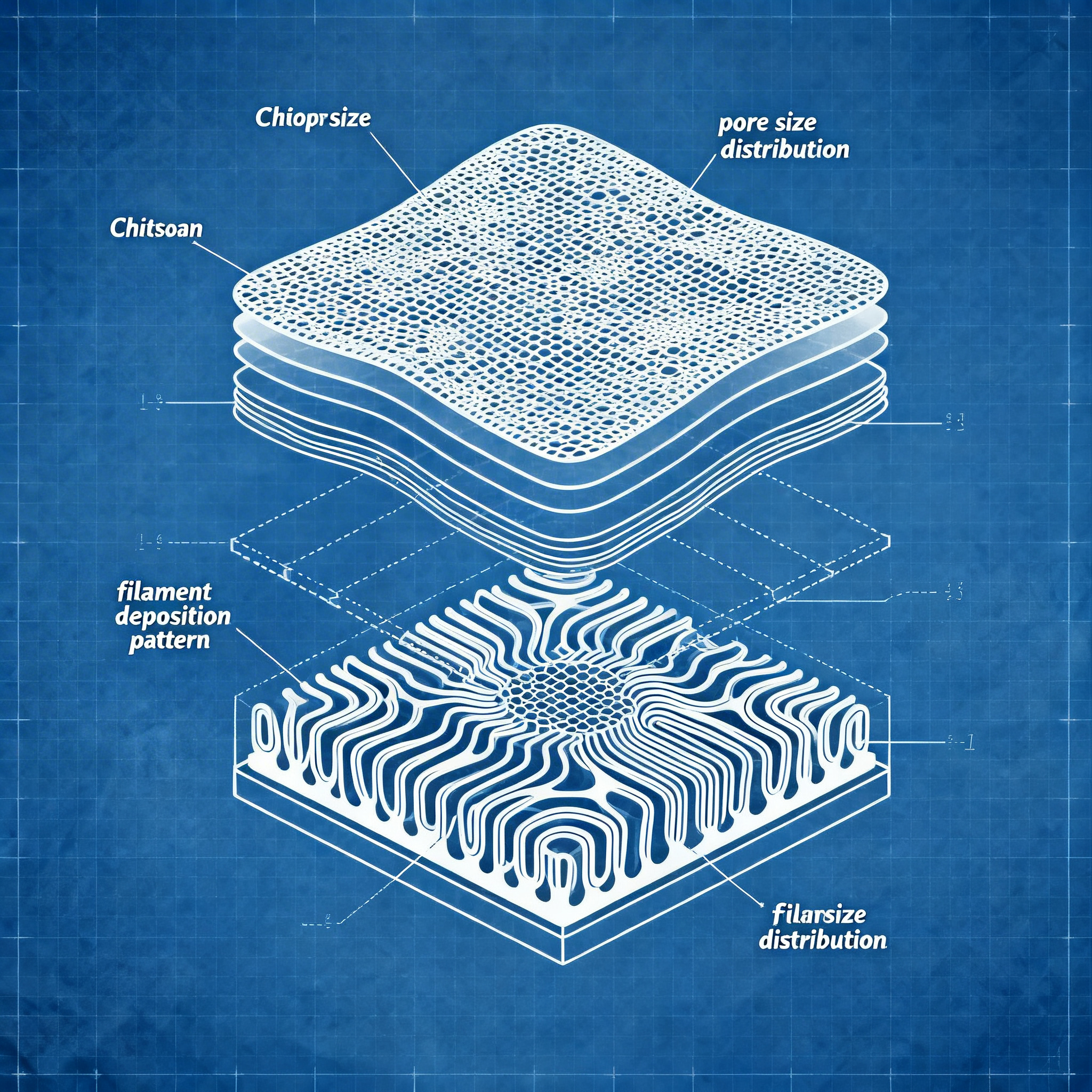
The base material chosen by the researchers is chitosan, a polysaccharide derived mainly from chitin found in the shells of crustaceans, insects, and some fungal species, already studied for biomedical applications and advanced dressings. Chitosan offers mechanical, antibacterial, and pro-regenerative properties that make it ideal for bioprinted scaffolds intended for wound healing.
Studies on chitosan-based dressings demonstrate acceleration of epithelialization, reduction of infections, and better quality of granulation tissue, in addition to good tolerability profiles even in prolonged applications. In experimental models of 3D-printed chitosan-based coatings, better organization of granulation tissue, an increase in the number of newly formed capillaries, and a reduction in local inflammation were observed compared to controls, confirming the regenerative potential of this biopolymer.
One of the advantages highlighted by the researchers is that the chosen materials are biodegradable: the scaffold is progressively absorbed by the skin, avoiding the need for traumatic removals and reducing the risk of toxic residues. PhD student Nouf Alshammari emphasizes that chitosan, as a material that is “inert” from a toxicological standpoint, reduces concerns related to systemic side effects, while maintaining a structural and functional role locally on the wound.
Bioprinting Technologies Applied to Skin Ulcers
3D printing allows for the geometric and functional customization of devices, adapting them exactly to the morphology of the lesion and enabling precise control of the scaffold's internal architecture.
At the core of the project is a porous scaffold, 3D printed, which can be designed in terms of shape, size, and internal architecture to adapt to the geometry of different wounds and different body areas. The structure is conceived as a “smart patch” that allows the passage of oxygen and moisture, essential for the healing of chronic ulcers, and at the same time hosts antimicrobial active ingredients capable of reducing the bacterial load locally.
The use of 3D printing allows the University of Mississippi to design “custom-made” patches: theoretically, a hospital ward could generate the digital model of the wound from 3D scans and print an adapted patch on-site exactly to the patient's morphology. Additive manufacturing also facilitates the control of the distribution of active ingredients within the scaffold, for example by creating concentration gradients or different functional layers.
3D printing allows to modulate parameters such as thickness, pore density, and deposition patterns, obtaining more flexible patches for joint areas or more robust ones for pressure areas. In perspective, military units or emergency centers could use portable generators and compact bioprinters to produce chitosan-based patches directly on-site, adapting the formulation to the type of wound and the infectious risk of the context.
Multilayer Design and Pharmacological Release Control
It is possible to integrate active ingredient gradients or specialized layers to control the release and improve therapeutic efficacy, without resorting to traditional antibiotics that could favor bacterial resistance.
Beyond the intrinsic properties of chitosan, the University of Mississippi team integrates plant-derived antimicrobial compounds into the patch to increase efficacy against a broad spectrum of bacteria without resorting to long-term traditional antibiotics. According to Michael Repka, this approach also stems from the desire to avoid organic solvents in production, which can interfere with the healing process, and from the need to reduce the risk of onset of bacterial resistance associated with the continuous use of antibiotics.
Additive manufacturing facilitates the control of the distribution of active ingredients within the scaffold, as demonstrated in other works on “sandwich” dressings with internal layers loaded with antibiotics or pro-angiogenic molecules such as L-arginine. Parallel research in the academic field explores various combinations between chitosan and additive technologies to create advanced dressings. Some groups have developed antibacterial wound coatings based on chitosan and polyvinylpyrrolidone printed in 3D, loaded with povidone-iodine and dermal cells, obtaining high adhesion to the wound, a porous structure with parallel channels, and a marked effect on the speed of epithelialization.
Other studies propose hydrogels based on amphiphilic derivatives of chitosan or on chitosan-cerium oxide composites printed in 3D, capable of counteracting both infections and oxidative stress in chronic wounds, paving the way for increasingly customizable multifunctional dressings.
Clinical Implementation Process: From Diagnosis to Printing
A clinically oriented workflow allows for the transition from the acquisition of the wound morphology to the production of the personalized therapeutic device, with the goal of integrating bioprinting into standard care pathways.
The concept of the patch is designed primarily for chronic ulcers due to diabetes, venous insufficiency, pressure sores, or other conditions that compromise microcirculation. In these cases, the combination of mechanical support, antibacterial effect, and moisture regulation creates a microenvironment more favorable to wound closure, while simultaneously reducing the risk of bacterial biofilms that often hinder healing.
Clinical trials on chitosan-based gels or dressings in patients with diabetic foot ulcers already indicate a benefit in terms of reduction in lesion size and healing times compared to standard therapies, an element that supports the rationale for an evolution towards more sophisticated 3D-printed systems.
Before large-scale clinical adoption, in-depth preclinical and clinical studies will be necessary to evaluate safety, efficacy, material stability, storage management, and integration into hospital workflows, but data available so far on similar systems show promising signals in terms of infection control and improvement of tissue regeneration.
Conclusion
The combination of bioprinting and chitosano represents today an innovative and clinically applicable solution to respond in a targeted way to the needs of chronic ulcers.
In the medium term, the combination of biopolymers such as chitosan, natural antimicrobial agents, and additive printing and bioprinting techniques could become a key platform for customized dressings intended for the most complex forms of chronic ulcers. Advanced healthcare centers can now consider the integration of local bioprinting systems to optimize therapeutic pathways related to regenerative medicine, reducing intervention times and improving clinical outcomes for the most fragile patients.
article written with the help of artificial intelligence systems
Q&A
- What are the main characteristics of chitosan that make it useful for the healing of chronic ulcers?
- Chitosan is biocompatible, biodegradable, and promotes cell adhesion. It possesses intrinsic antibacterial properties, accelerates epithelialization, and improves the quality of granulation tissue, reducing the risk of infections.
- How does the 3D-printed chitosan-based patch work?
- The patch acts as a porous 'scaffold' that supports tissue regeneration, releases natural antibacterial agents in a controlled manner, and maintains a microenvironment favorable to healing, adapting exactly to the shape of the wound.
- What advantages does 3D printing offer in the production of these patches?
- 3D printing allows for the exact customization of the patch based on the wound morphology, precise control of the internal architecture, and targeted distribution of active ingredients, improving therapeutic efficacy and adapting to different body parts.
- Why do researchers avoid the use of traditional antibiotics in their patches?
- Traditional antibiotics can promote the emergence of bacterial resistance. For this reason, researchers use natural antimicrobial compounds that offer a broad spectrum of action without this risk.
- What types of ulcers can benefit from this technology?
- This technology is particularly indicated for chronic ulcers due to diabetes, venous insufficiency, bedsores, or other conditions that compromise microcirculation and delay natural healing.